No. 19 Development of versatile deodorizing agent using cyclodextrin-iodine complex (CDI)
Summary
Iodine complexes out of β-cyclodextrin (β-CD) and chemically modified β-CDs are now industrially available. Iodine – methylated β-CD complex (MCDI) was prepared as a water-soluble deodorization agent. It was shown that all of those iodine complexes have an excellent deodorization effect against broad range of smelly volatile compounds.
Experimental method of deodorant effect
Experimental method
Each testing material 5 g and each odor ingredient are added into the bag (poly-fluorovinyl) containing 3 L of air, and gas concentration is analyzed at the fixed time by a gas-detector.
Samples
BCD (β-CD)
BCD-I-20 (Iodine / β-CD complex, Available iodine content 20%)
MCD-I-3 (Iodine / methylate β-CD complex, Available iodine content 3%)
PVP-I (Polyvinylpyrrolidone / Iodine complex, Available iodine content 10%)
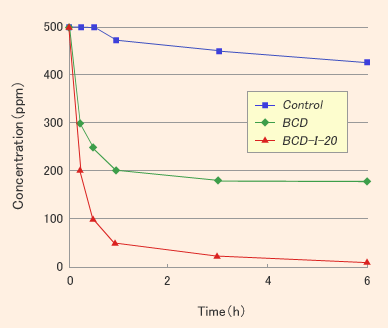
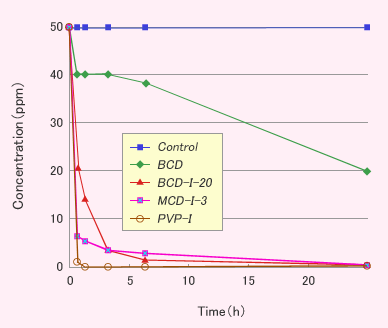
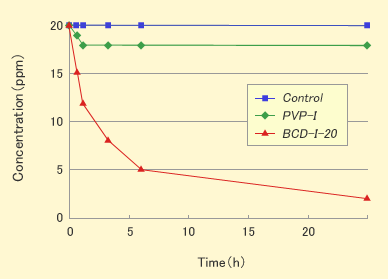
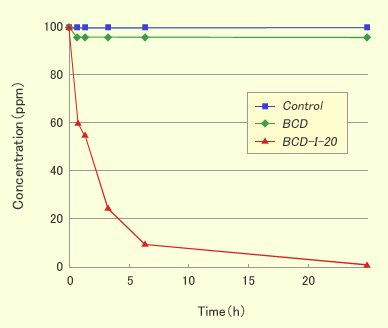
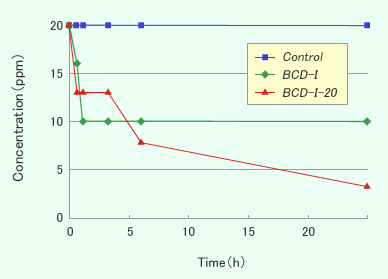
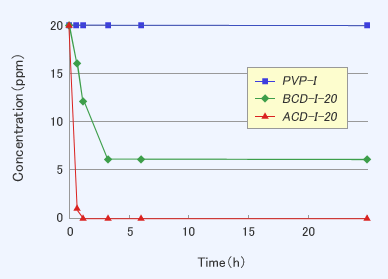
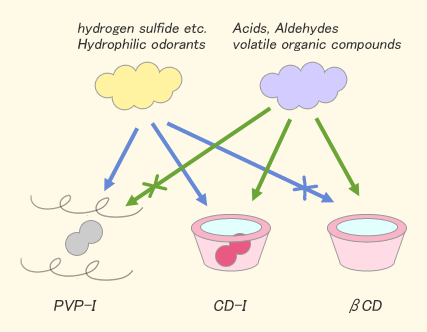
Results and Discussions
- Iodine showed the deodorant effect against some nitrogen compounds like ammonia and trimethylamine with / without existence of CD or PVP.
- β-CD did not show the deodorant effect against hydrogen sulfide as a hydrophilic compound but CDI showed.
- CDI showed the deodorant effect against methyl mercaptan, but PVP-I did not show.
- CDI showed the deodorant effect against some volatile organic compounds like aldehydes or acids, but PVP-I did not show. The results mean that the deodorant effect was originated from the interaction between organic compounds and the hydrophobic cavity in CD.
