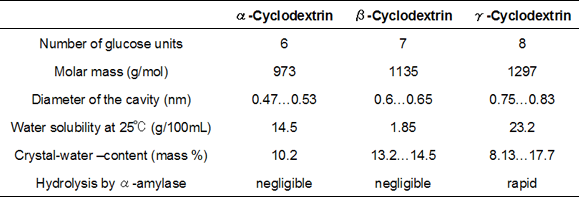
CDs are non-reducing, chiral cyclic oligosaccharides in which D-(+)-glucopyranose units are linked α-(1,4)-gylcosidically into a ring. Depending on the number of D-(+)-glucopyranose units - and thus also on the size of the ring - a distinction is made among α-CD, β-CD and γ-CD: α-CD consists of six glucose units, β-CD of seven and γ-CD of eight.
CDs are natural conversion compounds of starch, which is a polymer of glucose. In nature, starch is enzymatically decomposed by specific microorganisms into CDs and saccharide chains. Highly active bacteria that can selectively produce oligosaccharide rings have been selected by combining modern and refined techniques. *1 The enzyme for producing oligosaccharide ring has a formal name of 1,4-α-D-glucan 4-α-D-(1,4-α-D-glucano)-taransferase (cyclizing), and it is also called cyclodextrin glucosyl transferase or merely CDTase. A research group in Freigburg succeeded in crystallizing the enzyme and reported the crystal structure of CGTase. *2
CDs have a toroid molecular structure, and the inner wall has a high has a high electron density due to the arrangement of hydroxyl groups and is hydrophobic. On the other hand, the outer wall is hydrophilic. (Fig 1)
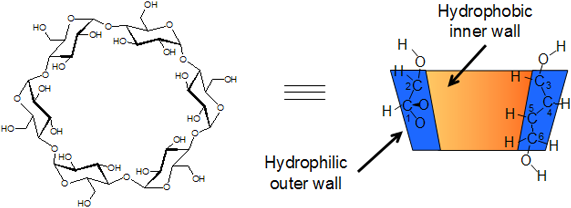
The secondary hydroxyl groups are located at the larger opening of the toroid. The primary hydroxyl groups, which are located at the smaller opening, are rotatable in a manner that they can change the size the opening. These hydroxyl groups determine the water solubility of CDs. The height of the cavity is 0.8 nm, and its diameter depends on the size of the ring. (Table 1)
CDs are slightly sweet white powder, are highly more stable than starch and sucrose, and can be stored over years under appropriate conditions.
A characteristic feature of CD molecules is their ability to enclose lipophilic substance reversibly in their hydrophobic cavity, provided the guest molecule size and geometric shape enable it to fit in the cavity. These CD inclusion compounds, which are known as host-guest complexes, are held together by Van der Waals' forces.
1 Schmid G, Huber O S, and Eberle H J, Selective complexing agents for the production of γ-cyclodextrin, Proc. 4th International Cyclodextrin Symposium, 87-92 (1988)
2 Schmid G, Cyclodextrin Glycosyltransferases and their Impact on Production ofCyclodextrins. 29th Cyclodextrin Symposium (2011)
